Investor News
'Green' steel articles omit cost
- 'Green' steel applications are being developed by a range of companies
- 'Green' steel is the term used to describe a production method that uses 'renewable hydrogen' instead of coking coal, eliminating CO2 emissions
- 'Renewable hydrogen' is made using electricity generated by wind turbines and solar panels
- Media reports routinely leave out discussion of cost, which is essential to understanding economic feasibility
- According to BNEF, a pro-renewable research organisation, 'green' steel becomes economical when the hydrogen cost drops below USD 2.20kg (AUD 3.25kg) and coking coal rises above USD 310 per tonne (AUD 435t)
- Renewable hydrogen generation in Australia is estimated to drop in cost from AUD 11.00kg to AUD 6.79kg by 2030, highlighting the need for a significant but as yet unknown breakthrough for 'green' hydrogen and 'green' steel to become economic
- Our HydroMOR process is an alternative, lower-cost, hydrogen-based iron-making process currently under development that can help bridge the gap between today's CO2-intensive steelmaking methods and tomorrow's zero-emissions solutions
An article in today's 'Manufacturers Monthly' highlights an AUD 30 million Austrian pilot project to demonstrate steel production using hydrogen instead of metallurgical coal.
The worlds largest steelmaker Arcelor Mittal is also building an AUD 105 million demonstration plant in Hamburg, Germany.
Swedish steelmaker SSAB has embarked on a program to develop the 'Hybrit' process. The program aims to deliver a 1-2 tonne per hour pilot plant at the cost of AUD 210 million and scale to commercial capacity by 2035 at an estimated cost of 20 to 30 per cent more per tonne of steel than existing methods.
In each case, the aim is to produce hydrogen using electricity generated by wind and solar, then use that hydrogen to convert iron ore to iron with no CO2 emissions.
Reading through the Manufacturers Monthly article, it quickly becomes apparent that the most important factor is omitted: cost.
Omitting cost has become a regular and concerning occurrence across the media, giving readers the impression that affordable 'green' hydrogen and, by extension, 'green' steel are easily attainable and just around the corner.
It isn't.
A 'green' steel industry based on 'green' hydrogen is a long way off.
Here's why.
The reason comes down to the physics and chemistry of steelmaking.
The dominant steelmaking process globally is the blast furnace route, which uses coke made from metallurgical grade coal. This entails a carbon-based chemical reaction at around 1300-1500 degrees Celsius temperatures.
Iron is abundant in nature and is mostly found combined with oxygen to form various iron ores. The two main commercial iron ores are hematite (Fe2O3) and magnetite (Fe3O4).
The chemistry of reducing iron oxide to iron basically involves a reaction between the carbon (in the form of carbon monoxide, CO) from the coke and the oxygen in the iron ore, leaving iron and making CO2, represented by the following equations.
Magnetite:
Hematite:
On average, each tonne of steel produced via the traditional blast furnace route emits 1.8 tonnes of CO2, with the steel industry accounting for about 7 to 9 per cent of direct emissions from fossil fuels.
Steel is simply iron, further refined to certain parameters via the addition or removal of particular elements, such as carbon, nickel and chrome, to produce various grades of steel.
Steel can be formed into various products, such as bars, rods, sheets, and slabs, ready for manufacturers to make the products we consume.

If hydrogen replaces coke, the main emission is H2O, which is water vapour.
The 'green' steel route looks like this:
The chemistry of hydrogen-based iron oxide reduction looks like this...
Hematite is converted to iron (Fe) in two steps, first to magnetite, then to iron:
3Fe2O3+H2 -> 2Fe3O4
Fe3O4+4H2 -> 3Fe+4H2O
Magnetite (Fe3O4) is converted to iron (Fe) in one step.
Sounds relatively straightforward.
However, steelmaker Arcelor Mittal has acknowledged that industrial scale-up is likely to take 10-20 years, and the cost of the steel is projected to be some 60 to 90 per cent higher than existing methods, calling into question not only the timeframes but fundamental commercial feasibility.
Steel is a globally traded commodity, and with over half the world's production coming from China, competitive pricing is non-negotiable.
As such, the main barrier for 'green' steel seems to be the cost of producing 'green' hydrogen.
Current modelling estimates that 51kg of hydrogen is required to make 1 tonne of steel.
Here in Australia, the cost of 'green' hydrogen is estimated at about AUD 11.00kg and is forecast to drop to around AUD 6.79kg.
This is well above the USD 2.20kg (AUD 3.25kg) level suggested by Bloomberg New Energy Finance (BNEF) as the economic threshold for feasibility.
This is a huge economic gap with no obvious technical solution or breakthrough on the horizon.
This is where our HydroMOR process comes in.
HydroMOR stands for 'hydrogen metal oxide reduction,' and it takes a unique approach that replaces coke, reduces CO2 emissions by around 35% compared to the blast furnace route, and is estimated to be cheaper than the dominant blast furnace method today. It's not reliant on the coking coal price, which reached USD 310 per tonne in 12 years, to be competitive, as BNEF suggests in the case of 'green' steel.
HydroMOR achieves this through the clever use of lower-cost, abundant, alternative raw materials and an innovative chemical and process pathway.
We can produce a composite pellet by combining lignite (brown coal) with iron ore fines. Our Coldry technology performs the pelletisation process, efficiently removing most of the moisture from the coal during the process.
The composite pellets are charged to a proprietary furnace and heated.
This is where the 'magic' happens.
Unlike coking coal, lignite is rich in volatile matter consisting of organic hydrocarbons.
HydroMOR turns those hydrocarbons into gas, cracking the hydrocarbon gas to free the hydrogen, which in turn reacts to strip away the oxygen from the iron ore, leaving pure iron (Fe). Because all this happens from the inside of the pellet, the process is relatively quick and very efficient.
HydroMOR reduces iron ore to iron via a hydrogen reaction without the need for external hydrogen generation or storage, creating a direct reduced iron pellet, pictured below.

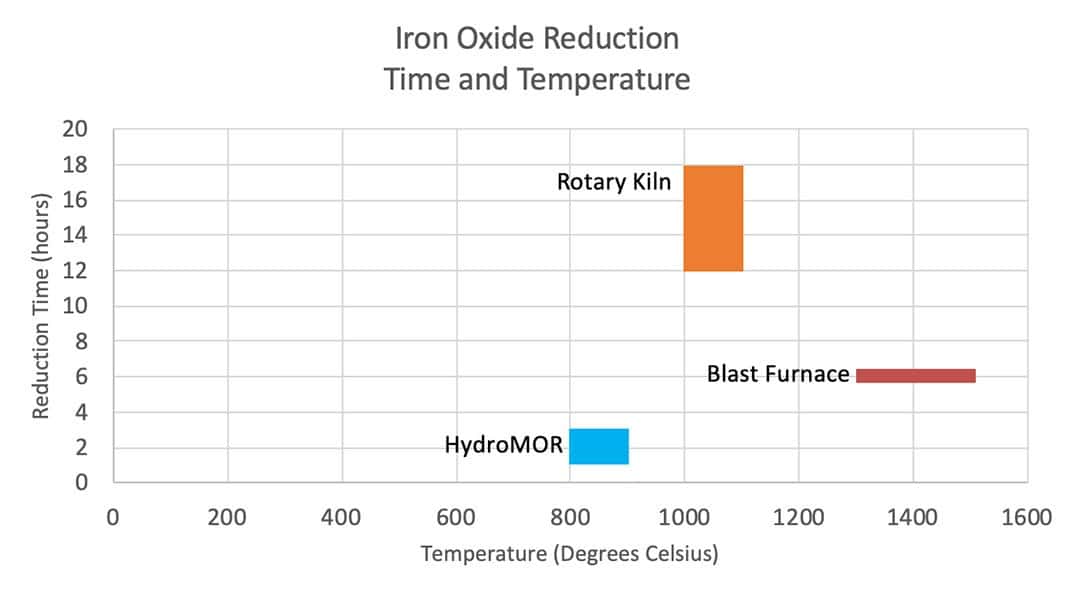
Further, HydroMOR achieves reduction at a lower temperature than the blast furnace or rotary kiln routes and in significantly shorter timeframes.
HydroMOR can also process iron ore fines or mill scale, which are considered lower-value than premium lump iron ore and often considered waste.
So, as you read more about 'green' steel and 'renewable' hydrogen, look for mention of the estimated production costs. If the cost is omitted, ask, 'What are they hiding, and why?'